Hearing the word “audit” is enough to send many professionals into a state of panic. That said, audits are not only necessary, but they are also vital to facilities that utilize cleanrooms. While facility managers may find audits stressful, this test of their personnel, protocols, and systems can bring to light issues that could ruin research. If you are coming up on a scheduled ISO 14644 certification audit or have had FDA inspectors drop by unexpectedly, you want to know your facilities are ready for an audit.
While you can control your protocols, personnel, and research methods, you can’t always control services you’ve outsourced, such as your cleaning service. Unfortunately, if your facility isn’t compliant with FDA and ISO cleanroom requirements, it won’t matter who did the work—your facility will fail the inspection.
To avoid these costly failed inspections, you will need to ensure your cleaning partner isn’t just a janitorial service. They need to fully understand the regulations you must follow, and they need to take an audit-ready approach to cleaning. Here at SourceONE Building Maintenance, we do more than just make certain your cleanrooms and other facilities are spotless—we keep detailed records and other reports necessary for an audit. Let’s take a look at what it means to be an audit-ready facility.
What Inspectors Look For
When an inspector walks into your biotech facilities, they’re not looking for dusty shelves or dirty floors, although they will certainly take note of those. What they’re really looking for are signs that your environments are controlled and can be repeatedly cleaned and controlled to specific standards. They often look for four specific factors:
1. Standardized SOPs
Your facility should be ruled by your Standard Operating Procedures or SOPs. These are your guiding principles, and inspectors are going to want to see that you have correctly written them and are following them closely. They want to see that you’re using the correct disinfectants and that floors are cleaned regularly. If the inspector asks three different people the same question, they are looking to see if they get the same answer. If they don’t, it’s a sign that your SOPs are not being applied consistently, and that’s a major issue.
2. Documentation
To the FDA, documentation is king. If something wasn’t properly documented, FDA inspectors will often assume it didn’t happen. They are going to scrutinize all of your cleaning logs to look for gaps, missed activities, missing signatures, and anything else that could indicate a lapse in protocol. They will make certain your documents clearly reflect your SOPs and that every cleaning cycle is clearly documented in chronological order.
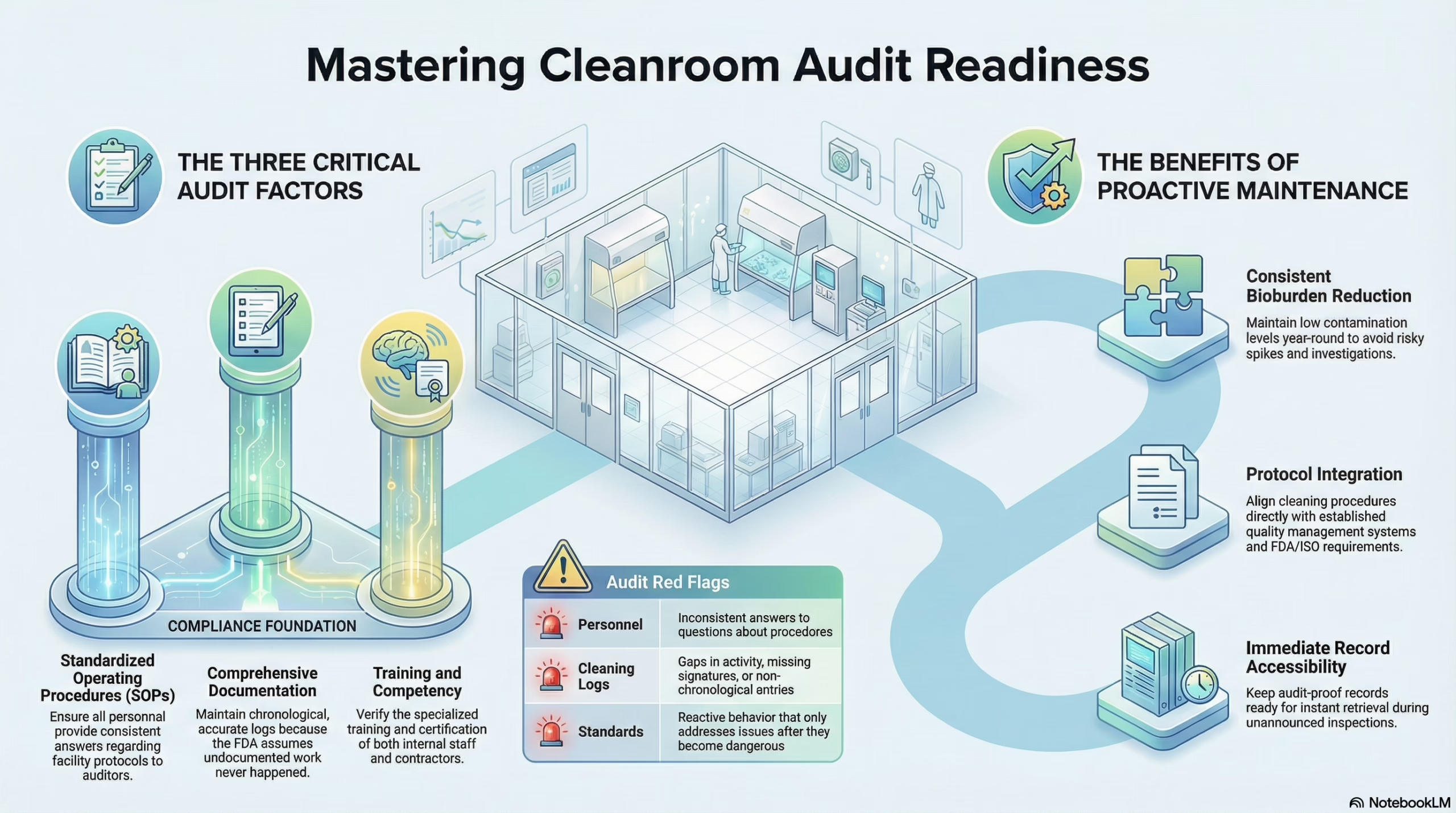
3. Training and Competency
While investing in the right facilities, equipment, and materials is important, none of it matters if you don’t have highly trained and competent people performing your research and managing your programs. This includes selecting the right cleaning service. Inspectors won’t stop at looking at what your own people do – they’re going to check your contractors, too. This is why you want to work with a service that fully trains their employees, provides documentation for that training, and verifies it regularly through exams and follow-up courses.
4. Continual Improvements
ISO and FDA standards evolve and change over time, and you need a cleaning team that pivots when needed. In addition to incorporating new standards, inspectors want to see that your cleaning team can identify trends in your environments, react to them correctly, and implement new procedures to acknowledge these trends. They want to see a proactive approach, not a reactive one that only addresses issues when they become a danger to the project.
The Benefits of Proactive Cleaning
Some facility managers focus on cleaning for the audit rather than cleaning for the sake of having a sterile, clean environment. A proactive cleaning program that focuses on creating the environment your team needs year-round is a better approach. Your facilities will be audit-ready, plus you will identify, document, and address any issues that could cause an audit failure.
When you take a proactive approach to cleaning, it becomes a part of your facility’s foundation. You reduce bioburden levels and keep them consistently low, reducing the risk of a spike in bioburdens that can require an investigation. Proactive cleaning also ensures that your documentation is up to date. If an auditor wants to see the last six months of cleaning logs, you won’t have to go digging for them. You or your cleaning service manager will be able to provide them immediately.
SourceONE Is Here to Help You Avoid Audit Issues
At SourceONE, we understand that as the facility manager, you’re often at the center of a company’s audit. That’s why we’re here to service not only as your cleaning services provider but also as an insurance policy for audits. We specialize in providing services to facilities where compliance is mandatory.
When don’t just clean your buildings—we integrate your protocols and quality management systems into our procedures. This ensures that every cleaning log, standard operating procedure, and process aligns with FDA and ISO cleanroom inspection requirements.
One of the benefits of working with us is that we take the burden of documentation off of your internal team. Our cleaning logs are designed to be audit-proof. To that end, we strive to keep logs that are accurate, chronological, and clear. When you’re asked for proof of compliance for an audit, our records will provide you with a transparent and detailed list of all activities done.
All of our technicians are trained for cleanroom environments, including understanding airflow patterns, gowning protocols, and cleaning flow. We’re not just a contractor; our team acts as an extension of yours, following all of your protocols and instructions.
Be Audit-Ready with SourceONE
The worst time to realize your cleaning protocols aren’t up to regulation is during a surprise audit. Being prepared is the only way to avoid a failed inspection or certification. Fortunately, that’s where Source ONE can help. When you partner with us, we will make certain your cleaning logs are up to date, your current operating procedures reflect the industry’s best practices, and that your facility is ready to be inspected by even the toughest auditor.
With our history of supporting successful audits, you can think of us as your secret insurance policy against inspections. To that end, we’ve prepared a free facility assessment and audit-readiness checklist that can help you evaluate gaps before your next audit. With a list of these areas that may need improvement, you can shift from a reactive to a proactive approach to cleanliness. Whether your next audit is six months out or six days away, partnering with SourceONE can help you move forward with confidence.
You can’t ignore compliance, especially in cleanrooms and similar facilities. Reach out to SourceONE today to learn how we can help and to schedule your assessment.